|
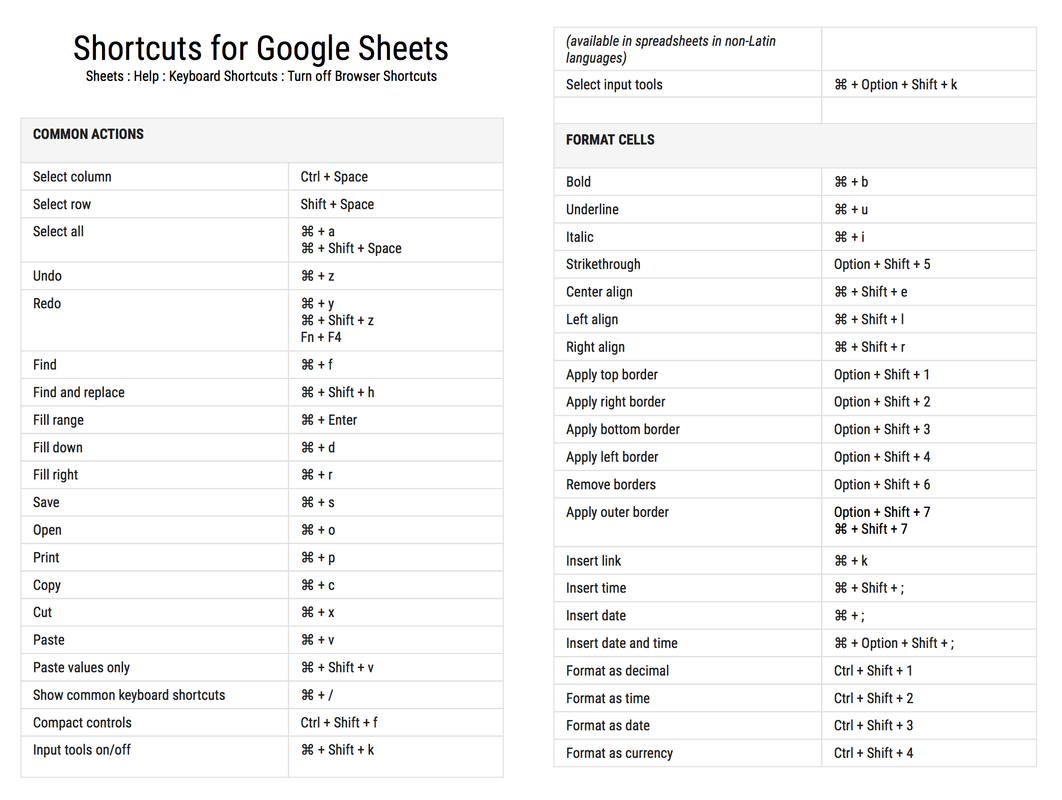
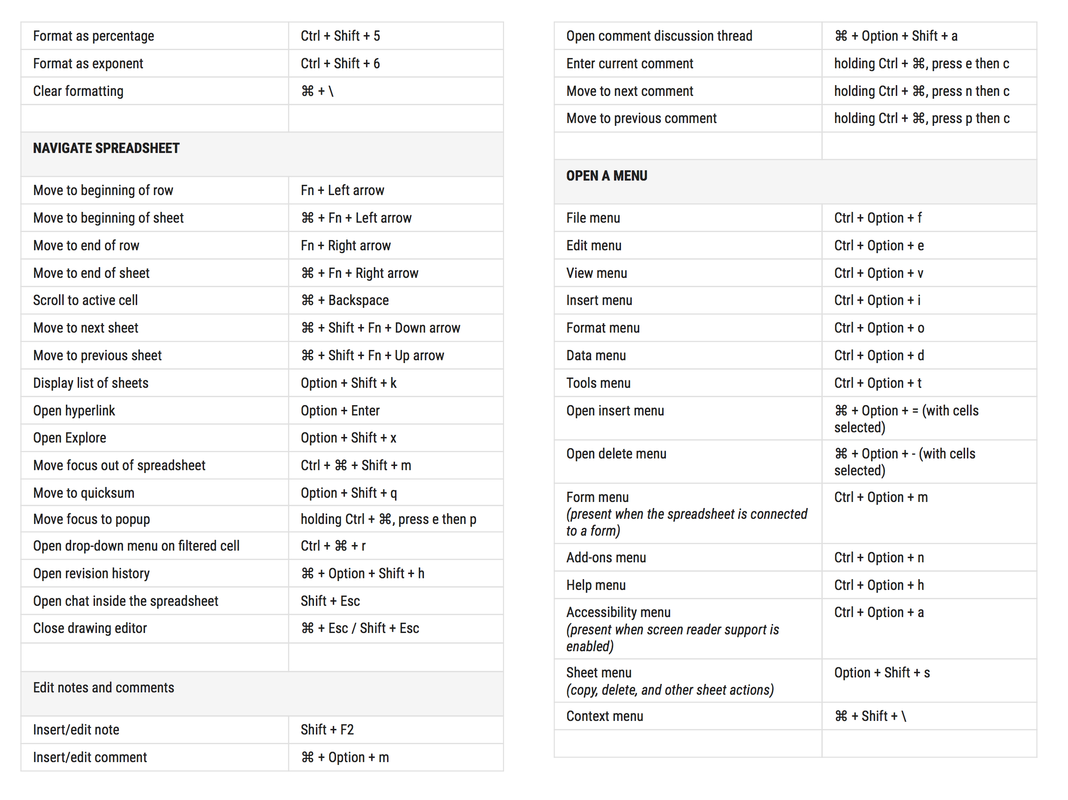
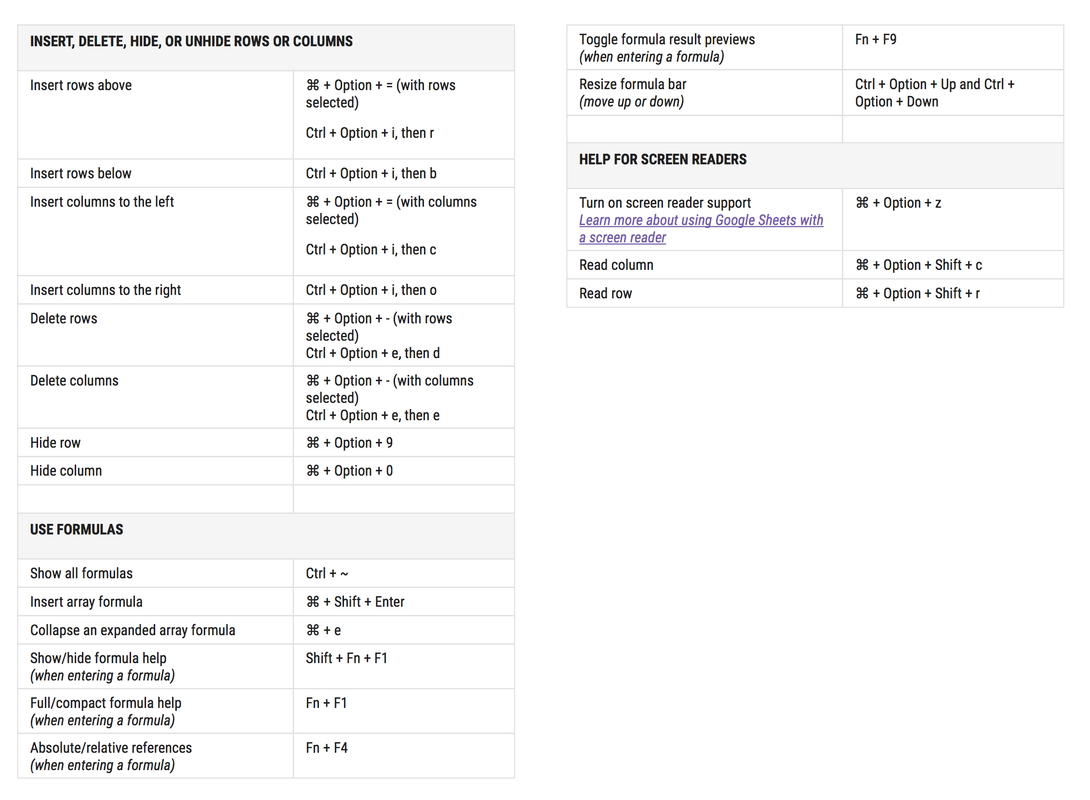
To activate these keyboard shortcuts:
*
How much carbon dioxide is produced when different fuels are burned? Different fuels emit different amounts of carbon dioxide (CO2) in relation to the energy they produce when burned. To analyze emissions across fuels, compare the amount of CO2 emitted per unit of energy output or heat content. Pounds of CO2 emitted per million British thermal units (Btu) of energy for various fuels: The amount of CO2 produced when a fuel is burned is a function of the carbon content of the fuel. The heat content, or the amount of energy produced when a fuel is burned, is mainly determined by the carbon (C) and hydrogen (H) content of the fuel. Heat is produced when C and H combine with oxygen (O) during combustion. Natural gas is primarily methane (CH4), which has a higher energy content relative to other fuels, and thus, it has a relatively lower CO2-to-energy content. Water and various elements, such as sulfur and noncombustible elements in some fuels, reduce their heating values and increase their CO2-to-heat contents. https://www.eia.gov/tools/faqs/faq.php?id=73&t=11 *
Here is a wonderfully succinct and apprehensible explanation of entropy — which I learned was nothing more than the probabilistic inevitability of macroscopic systems to approach a state of homogeneity, posited by the second law of thermodynamics.
In other words: "The mystery is, at the level of atoms and molecules, each of these processes are reversible. But when we get to bigger collections of atoms, a kind of one-way street emerges — a macroscopic irreversibility arises from microscopically reversible parts. Things spontaneously happen in the direction of increasing entropy, never in the opposite direction. "Now we know why. There’s no microscopic law telling any particle which direction to go, just like there’s no shepherd telling the sheep where to go in our imaginary farm. It’s just that there are more ways to spread energy around, and fewer ways to keep energy confined. Increasing entropy is highly likely, decreasing it is basically impossible. It’s just stuff obeying the laws of chance." *
|
(RE)SOURCESHere you will find a collection of material, ranging from technical data to white papers to theory, which has influenced my thinking. Archives
December 2020
Categories
All
|
|||||||||